Passwater:Good to chat with you again. Long-time readers may remember the last time we discussed your research back in 2007. You have been very busy in the meantime as your health research and clinical results are becoming of greater interest to health professionals everywhere. You continue to be the chief medical officer at the Riordan Clinic, in Wichita, KS. Please tell our readers a little about the famous Riordan Clinic.

Hunninghake:The Riordan Clinic is one of the largest non-profit, nutritional medicine centers in the United States. It was founded in 1975 by the late Hugh Riordan, M.D., a pioneer in nutritional medicine. We have a staff of about 40 people, including six full-time practitioners in addition to researchers and nurses. We have an analytical laboratory on-site for measuring vitamin and mineral levels as well as conventional medical tests. The physical structure of the clinic is also distinctive; it consists of eight geodesic domes and a pyramid. All of this is on a 90-acre nature preserve. Readers can find out more about the clinic at www.riordanclinic.org. I’m board certified in holistic medicine, and I’ve been practicing nutritional medicine at the center for almost 30 years.
Passwater:Dr. Ron, what piqued your interest in vitamin C?
Hunninghake:I was fortunate to be mentored for over 15 years by Dr. Hugh Riordan. In the early ‘90s, Dr. Riordan picked up the fallen research flag of Dr. Ewan Cameron, a Scottish physician, and Dr. Linus Pauling, a two-time Nobel Prize laureate. Dr. Cameron completed, and Dr. Pauling defended, clinical research utilizing high dose vitamin C for cancer. Cameron’s and Pauling’s efforts to demonstrate the cancer treatment benefits of vitamin C were thwarted by the Mayo Clinic’s incorrect attempt to replicate its exciting findings. The oncological community gave the nod to the more prestigious Mayo Clinic, and further efforts to investigate vitamin C were stymied…until Dr. Riordan came along.
Passwater:Sounds like a tragedy similar to what happened to the late Dr. Fred Klenner’s clinical findings.
Hunninghake:It was amazing for me to learn that Dr. Klenner, a respected graduate of Duke Medical School, had published a research series in the Journal of the American Medical Association (JAMA) where he reported curing 60 out of 60 cases of polio in the late 40s. He also presented his paper at an AMA meeting. The astonishing response: zilch, zero, no interest!
Passwater:I have always been very upset that his findings weren’t widely disseminated. I personally lived through the fear of the great polio epidemic when we were discouraged from congregating in large numbers, or using public swimming pools for fear of polio. We could barely build enough iron lungs to handle the devastating consequences of this horrible epidemic, yet a 100% cure was ignored!
There is still almost total ignorance of the work of Dr. Klenner and others with vitamin C against viral diseases and cancer.
Hunninghake:One encouraging statistic I’ve heard is that more people now have vitamin C in their medicine cabinets than aspirin. Even so, both the public and the medical profession are largely ignorant about vitamin C as an anti-microbial and detoxification agent. Dr. Thomas Levy has written a wonderful book, Vitamin C, Infectious Disease, and Toxins. He makes the very important point that DOSAGE is the key to vitamin C’s effectiveness. Everyone is different; each illness is different; severity is a factor; the individual’s diet, lifestyle, sleeping habits, smoking habits, concurrent meds, and overlapping diseases all make a difference in dosing! Most cases of vitamin C “failure” are due to improper, inadequate dosing. Sometimes, intravenous C is necessary to achieve the effective dosage threshold.
Passwater: Are these amazing medical benefits of vitamin C taught in medical schools?
Hunninghake:I had three hours of nutrition in medical school. No, not three semester hours…a total of three hours one afternoon! We discussed beriberi, scurvy, rickets and several other single nutrient deficiency illnesses…then the instructor said not to worry about them because they so rarely occur here in America. The idea of sub-clinical deficiencies, or multiple overlapping low-grade deficiencies was never presented.
Passwater:What natural evidence do we have to substantiate the concept that vitamin C at higher than RDA doses might help to treat serious illness?
Hunninghake:Let me answer your question by starting with how much vitamin C animals need and make. First, let’s consider the human animal — us! Humans don’t make our own vitamin C, so we will use the RDA for humans (90 mg/d) and scale it to an average human’s body mass which equals 1 mg/kg/d. Dogs do synthesize vitamin C, but in an amount equaling 3 mg/kg/d. That would be equivalent to about 200 mg/d for a human. Pigs also synthesize vitamin C at 8 mg/kg/d which is equivalent to about 500 mg/d for a human. Goat s are amazing. Unstressed goats can make up to 190 mg/kg/d. You and I would need to take 14 one-gram vitamin C tablets to equal that. Then, unbelievably… an injured goat can synthesize a whopping 1300 mg/kg/d! That’s 100,000 mg a day for a human: a whole bottle of vitamin C tablets!
Passwater:Other primates are especially closely related to man. Do non-human primates consume the minimum C necessary to get by?
Hunninghake:Like humans, other primates do not make vitamin C. They lack the ability to express the genome that makes a liver enzyme called GLO (L-gulonolactone oxidase). GLO converts glucose into vitamin C. So, how much do they get in their diet? Spider monkeys get about 100 mg/kg/d. That would be 7,000 mg a day for a human. Gorillas consume 30 mg/kg/d. That’s about 2 grams, which is far above the measly 60 mg the RDA experts would suggest humans consume to prevent scurvy.
There’s a great quotation from Dr. Levy’s book that touches on this point: “This automatic ability to adequately step up vitamin C production in the face of stress explains why so many wild animals tend to live healthy for their entire life spans…and conversely, generally vitamin C-depleted human beings will typically spend at least half of their lifetimes coping with one or more chronic diseases.”
Passwater:What is the optimum dose of vitamin C for humans?
Hunninghake:Dr. Linus Pauling estimated that two grams a day would go a long way towards preventing the common cold in the majority of people. But that’s just a guess. Dr. James Jackson, the former head of The BioCenter Lab at the Riordan Clinic, has developed an innovative way for each person to take into consideration all the many variables that can affect their vitamin C need for the day. It’s called VitaCheck C strips. These are very similar to urinalysis test strips, with a small chemical pad at the end of a plastic strip. You wave the test pad through your urine stream and the color change corresponds to the vitamin C concentration in your urine at that moment. If it’s low, you need to increase your intake. Your need can vary greatly depending on your diet, your stress load, whether you smoke, how well you slept the night before, and whether you are fighting a cold or some other oxidant-producing illness.
Passwater:Vitamin C doesn’t seem to fit the standard definition of a vitamin does it?
Hunninghake:It depends on how you define “vitamin.” If you mean “a substance that must be provided by food or supplementation” then, yes, it definitely is a vitamin. If you take the medical definition that a vitamin is “a general term for a number of unrelated organic substances that occur in many foods in small amounts and that are necessary in trace amounts for the normal metabolic functioning of the body,” then vitamin C may not be a classic vitamin, because in large amounts it helps living organisms adapt better to stress, trauma, and/or illness. I believe we should stop referring to it as vitamin C. We should start calling it “adaptogen C.”
Passwater:Others have made strong cases for vitamin C being more than a vitamin. Even Dr. Albert Szent-Gyorgyi, who was awarded the 1937 Nobel Prize in Medicine for discovering vitamin C, saw ascorbic acid as being more than “anti-scorbutic.”
Hunninghake:Yes, Dr. Szent-Gyorgyi saw vitamin C as “The Electron Exchanger.” In his words, vitamin C is “… one of the primary substances assuring that a vigorous, continuing electron exchange takes place among the body’s tissues and molecules. One definition of life, then, is that it is a state in which an optimal degree of electron interchange among cells can take place.” This corresponds to what could be simply defined as three states of vitamin C intake: minimum (like the RDA); average (like the primates in the wild); and optimal (like the goats who can really go to town making huge amounts of adaptogen C when they really need it).
Passwater:The Center was founded by Dr. Hugh Riordan and you worked alongside him for a good while. What drew Dr. Riordan to IV vitamin C?
Hunninghake:Well, he was very aware of Dr. Klenner’s findings. But, there was a defining experience in his life that really captured his scientific attention: In 1975 Dr. Riordan was bitten by what he believed was a Recluse spider. When he checked his plasma vitamin C level – zero! “I’ll fix that,” he thought. He got one of the Center nurses to give him 15,000 mg of ascorbic acid IV. He rechecked his C level: still zero! Only after several IVCs did he resolve his temporary state of scurvy…some 50,000 mg of vitamin C later! A perfect illustration of the fact that the dosage of vitamin C an individual needs at any given time is totally dependent on several variables that impact their oxidative stress load…like a Recluse spider bite.
Passwater:Where did this lead him?
Hunninghake:Real estate agents have a pet saying: location, location, location! Dr. Riordan’s was: measure, measure, measure! After the spider incident he began to routinely measure plasma vitamin C levels on all chronically ill patients coming to The Center. You can almost guess what he found: sickness and low plasma C go hand in hand. And in cancer patients he found very low vitamin C reserves! When he checked the literature, he found that a low vitamin C status was a consistent finding in cancer studies where the researchers bothered to check plasma vitamin C levels.
Passwater:Why do cancer patients have such low vitamin C levels?
Hunninghake:The membranes of cancer cells contain large numbers of glucose transporters that seek glucose. Thus, cancer cells remove vitamin C from the blood by mistake in the cancer’s efforts to take in more glucose needed for fuel for its energy.
Passwater:Please explain further.
Hunninghake:The vast majority of living creatures make the vitamin C they need from blood sugar. As chronically injured cells sustain damage to their DNA, oxidative stress builds up. This increases their tendency to divide. Over time, if the damage is progressive and irreversible, the cells will lose their control mechanisms and begin to function more like single-celled organisms rather than cells that are part of a multi-cellular organism. This is the shift towards cancer, and cancer cells are anaerobic obligates… their metabolic pathways require glucose.
Passwater:OK. So, getting nutrients into the bloodstream is only part of the story. The nutrients must also get into the cells to do their jobs. There are transporting proteins embedded in cell membranes to carry specific nutrients into the cell interiors where they can do their work. Human cells produce glucose transporters, primarily various members of a “family”called GLUTs, depending on the cell. Vitamin C can enter cells directly and passively via diffusion, but vitamin C can also be actively transported into cells via GLUT, but moreso via Sodium-Ascorbate Co-Transporters (SVCTs).
The size, structure and electronic charges on the perimeter of vitamin C molecules so closely mimic those of glucose molecules that the glucose receptors undergo conformational changes that facilitate vitamin C entry into the cells. Please see Figure 1.
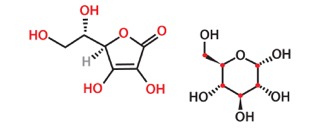
Most cancer cells have large numbers of glucose transporters because glucose is critical to cancer growth. So, the glucose transporters in the membranes of cancer cells, in their effort to bring more needed glucose into the cancer cells, mistakenly bring in vitamin C instead. Thus, vitamin C starts accumulating in cancer cells. What does this do?
Hunninghake:Recent research from the National Institutes of Health confirmed our Center’s findings from the early ‘90s: at very high dosages, vitamin C interacts with copper and iron to form the powerful hydroxyl radical. This free radical has a strong oxidative effect on cancer cells, which lack the protective enzyme catalase. Once the threshold dose of vitamin C was attained in cell culture, all types of cancer cells were selectively killed. Healthy, non-cancerous cells were spared…partly because of their much higher levels of catalase, and partly because they do not have near the same number of glucose (and vitamin C) transporters in their membranes as cancer cells.
Passwater:So high-dose vitamin C is cytotoxic to cancer cells, but cell-protective to non-cancerous cells? Amazing! How does the vitamin C kill the cancer cells?
Hunninghake:The same way chemotherapy and radiation do: severe oxidation. When cellular oxidative damage is severe enough, a special gene called the p53 tumor suppressor gene triggers apoptosis. Apoptosis is “programmed cell death.” In a multicellular organism, when cells are damaged beyond repair or worn out, the p53 mechanism simply causes the cell to self-destruct, for the greater good of the whole organism. In cancer cells, the p53 DNA of a healthy cell has been damaged and the cell has mutated. Cancer cells act like “selfish” single-celled organisms whose only intent is self-replication and survival…even if it means the death of the host organism.
Passwater:In 2017, you published the exciting results of the Riordan’s Clinic with IV vitamin C against prostate cancer. The published report concludes that your study “demonstrated the clinical benefit of IV vitamin C for prostate cancer patients“ (1). It is a seminal study that should open a lot of eyes. All physicians and cancer patients should know about it. The results are very important and encouraging for cancer patients. Your co-researcher in the study is Dr. Nina Mikirova of the Riordan Clinic. Let’s chat with Dr. Mikirova about this study next month. Dr. Hunninghake, thank you for discussing the basics of IVC against cancer with us and for your decades of research.WF
Reference1. Mikirova, N. and Hunninghake, R. “Changes in the rate of PSA progression and the level of alkaline phosphatase during high dose vitamin C treatment of patients with prostate cancer.” Functional Foods in Health and Disease 2017; 7(7) 511-528.
NOTE: The statements presented in this article should not be considered medical advice or a way to diagnose or treat any disease or illness. Dietary supplements do not treat, cure or prevent any disease. Always seek the advice of a medical professional before adding a dietary supplement to (or removing one from) your daily regimen. The opinions expressed in bylined articles are not necessarily those of the publisher.
Published in WholeFoods Magazine April 2018









