We have been discussing bone and artery health with Leon Schurgers, Ph.D. In June, we discussed why vitamin K2 was essential for bone health and in July, we examined why vitamin K2 is also essential for healthy arteries. Both involve keeping calcium in the bones and out of the arteries. Even Time magazine is now reporting that calcified arteries are a major risk factor in heart disease. New research indicates that there is more to the vitamin K2 story. Let’s continue our chat with Dr. Schurgers with a discussion about vitamin K2’s role in brain health.
Dr. Schurgers is a senior scientist in biochemistry at Maastricht University in the Netherlands. As a senior scientist for the division on vitamin K research, Dr. Schurgers is part of the Cardiovascular Research Institute, the largest research institute of the University of Maastricht. Dr. Schurgers’s major occupation is leading the scientific part of vitamin K research and supervising technical engineers, post-docs and Ph.D. students. Dr. Schurgers is a member of the American Society of Bone and Mineral Research, the International Society on Thrombosis and Haemostasis and the American Heart Association. He has published over 120 research papers in the international scientific press.
Passwater: The research on bone and artery health has been extremely interesting and is so important to everyone’s health, but there is even more to the vitamin K2 story. What can you tell us about research into Parkinson’s disease?
Schurgers: This is a very exciting new area of research from the  laboratory of neuroscientist Patrik Verstreken, Ph.D., of VIB (Flanders Institute for Biotechnology) and KU Leuven (the University of Leuven) in Belgium. VIB is a life sciences research institute in Flanders, Belgium, that has more than 1,300 scientists from over 60 countries. VIB works in close partnership with four universities: Ghent University, KU Leuven, University of Antwerp and Vrije Universiteit Brussel. Dr. Verstreken was awarded the 2014 International Brain Research Organization’s Kemali Prize for his research, which included investigating the effect of one genetic defect leading to Parkinson’s and found a major role for vitamin K2.
laboratory of neuroscientist Patrik Verstreken, Ph.D., of VIB (Flanders Institute for Biotechnology) and KU Leuven (the University of Leuven) in Belgium. VIB is a life sciences research institute in Flanders, Belgium, that has more than 1,300 scientists from over 60 countries. VIB works in close partnership with four universities: Ghent University, KU Leuven, University of Antwerp and Vrije Universiteit Brussel. Dr. Verstreken was awarded the 2014 International Brain Research Organization’s Kemali Prize for his research, which included investigating the effect of one genetic defect leading to Parkinson’s and found a major role for vitamin K2.
Dr. Verstreken specializes in mechanisms underlying presynaptic transmitter release, and its relevance to neurodegenerative diseases including Parkinson’s disease. The mitochondria in cells produce the energy needed by cells to function. Dr. Verstreken has identified vitamin K2 as a mitochondrial electron carrier molecule that is required to maintain high levels of mitochondrial activity and neurotransmitter release. These findings are important for understanding how nerve synapses function and have potential relevance to devising therapies for Parkinson’s disease and related disorders.
His discovery might result in a future treatment option for Parkinson’s patients that involves taking vitamin K2. As I mentioned, the mitochondria in the cell supply the energy needed for cells to function. This activity is disrupted in Parkinson’s patients, resulting in lower energy production, which causes brain cells to die and loss of neural communication. Over time, affected individuals experience lack of movement (akinesia), tremors and muscle stiffness. Parkinson’s patients have several genetic defects, including PINK1 and Parkin mutations that lead to reduced mitochondrial activity. Both the PINK1 and Parkin genes are involved in vitamin K2 metabolism.
In one of his studies, Dr. Verstreken and his colleagues gave vitamin K2 to fruit flies with a genetic defect in PINK1 or Parkin, and mitochondrial energy production and electron transport were restored. Vitamin K2 plays a role in the energy production of defective mitochondria. Because defective mitochondria are also found in Parkinson’s patients with a PINK1 or Parkin mutation, vitamin K2 potentially offers hope for a new treatment for Parkinson’s.
Passwater: And memory and cognitive function? What are the mechanisms here?
Schurgers: Vitamin K was identified some 40 years ago as being essential for the synthesis of sphingolipids. Sphingolipids are a complex and versatile family of lipids (fats) that play important roles in signal transmission and cell recognition. In addition to their structural role, these lipids have particular impact on neural tissue as they are present in high concentrations in brain cell membranes.
Vitamin K-dependent proteins are now known to play key roles in the central and peripheral nervous systems. Growth arrest-specific 6 (Gas6) is a gene that regulates Gas6 protein. Notably, Gas6 protein has been shown to be actively involved in cell survival, chemotaxis (i.e., the migration of neurons in response to a stimulus), mitogenesis (i.e., the induction of cell division), and cell growth of neurons and glial cells.
A recent study found that patients with early-stage Alzheimer’s disease consumed less vitamin K than control subjects who were cognitively intact. In 2001, A.C. Allison, D. Phil., hypothesized that vitamin K deficiency could contribute to the pathogenesis of Alzheimer’s disease, based on the potential actions of vitamin K in the brain and through a link to the apolipoprotein E genotype (1). The apolipoprotein E4 allele, an established risk factor for Alzheimer’s disease, is also associated with lower plasma vitamin K levels.
Moreover, increasing evidence suggests a role of atherosclerosis in the pathogenesis of cognitive impairment and dementia. Larger calcification volume is associated with worse cognitive performance. It also relates to smaller brain tissue volumes and worse white matter microstructural integrity, revealing possible mechanisms through which atherosclerosis may lead to poorer cognition.
This is a compelling area to explore, and more well-designed studies should be done to test such hypotheses.
Passwater: Agreed! Now, let’s review some of the more important differences between vitamin K1 and vitamin K2. Which factors are activated by both K1 and K2 and which are activated by K1 or K2 alone?
Schurgers: All K vitamins are similar in structure: they share a “naphthoquinone” ring structure. However, they differ in the length and saturation and number of attached carbon atoms in the side chain called, “isoprenoid units.” Please see Figure 1.
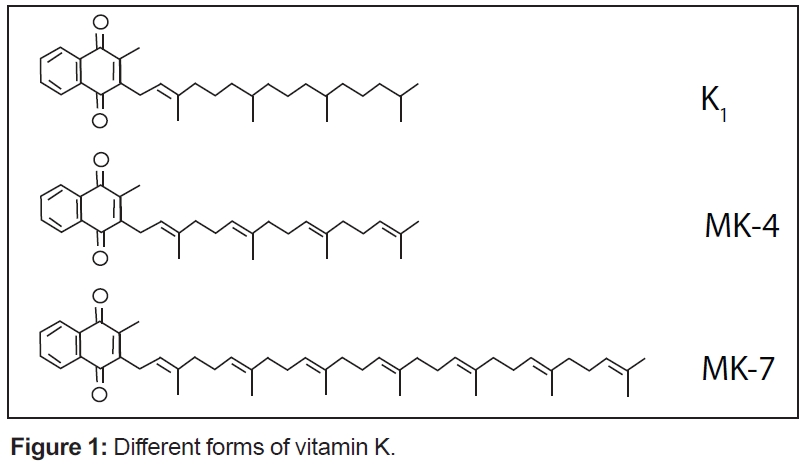
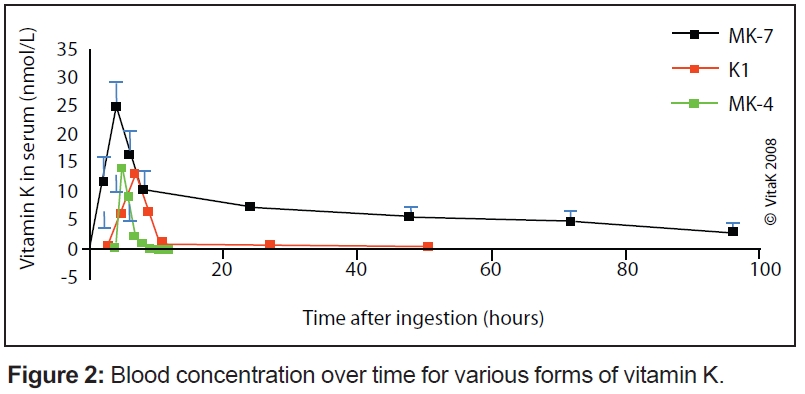
These differences are especially pronounced in the vitamin K2 group (also called menaquinones) with different lengths of the side chain. The length is marked in the name of the particular menaquinone chain (for example, MK-7 means that seven isoprenoid residues are attached to the ring structure). The length influences their abilities to reach different tissues within the body: the longer the side chain (seven to nine units) of the vitamin K, the better its potential due to half-life time in the blood. Consequently, the superior K vitamins are the long-chain menaquinones—especially MK-7, but to some extent also MK-8 and MK-9. They are completely absorbed as compared to vitamin K1, and because they are in the blood for longer time, they are more available for all tissues when needed (see Figure 2) Hence, these forms are also more active in tissues outside the liver such as bones, arteries and soft tissues; a small dose of MK-7 is required to fulfill the needs of the body (i.e., high bioavailability).
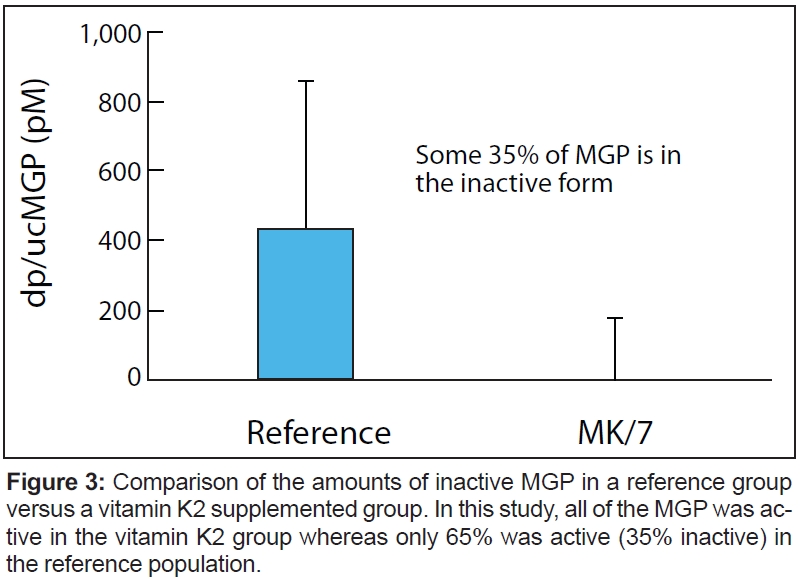
K vitamins (K1 and K2) play an essential role as cofactors for the enzyme gamma-glutamylcarboxylase, which is involved in carboxylation of the vitamin K-dependent proteins. This includes matrix Gla protein (MGP), which inhibits potentially fatal calcium accumulation in arteries. This means that the enzyme, with the help of the K vitamins, is responsible for making them biologically active. Without K vitamins, the reaction does not occur and the proteins that are synthesized stay biologically inactive. The major difference lies in bioavailability and bioeffectivity. Figure 3 compares the amounts of inactive MGP in a reference group versus a vitamin K2 supplemented group. In this study, all of the MGP was active in the vitamin K2 group whereas only 65% was active (35% inactive) in the reference population.
Passwater: In Figure 2, you show how much vitamin K2 reaches the bloodstream and continues in circulation compared with vitamin K1. Why is this important?
Schurgers: The comparison of vitamins K1 and K2 (MK-7) was published in the journal, Blood, in 2007 (2). In this paper, we compared the in vivo properties of two forms of vitamin K: MK-7 and K1. Both K1 and MK-7 were absorbed well, with peak serum concentrations at four hours after intake.
As shown in Figure 2, a major difference between the two vitamin K species is the very long half-life time of MK-7, resulting in much more stable serum levels, and the accumulation of MK-7 to higher levels (seven- to eight-fold) during prolonged intake. Moreover, after oral ingestion, MK-7 is more effective in both catalyzing osteocalcin carboxylation in bone and counteracting coumarin anticoagulants in the liver. The mechanism underlying this observation may be MK-7’s much longer half-life time in the circulation and its reported higher cofactor activity in vitro.
During recent years, many studies have demonstrated that the extra-hepatic vitamin K requirement exceeds the recommended daily allowance (100–120 mcg/d) for vitamin K1, which is set for the liver. For the food industry, an alternative to increasing the recommended dose would be introducing the more potent MK-7 instead of K1 on a larger scale in functional foods and multivitamin supplements.
Passwater: What is the optimal blood level for vitamin K?
Schurgers: Clinicians do not measure blood levels of K vitamins to check for deficiency. The main disadvantage is that circulating vitamin K levels correspond to the daily intake of green-leafy vegetables, cheeses, etc. The best way to evaluate vitamin K efficiency is to determine the decrease in the circulating undercarboxylated form of the vitamin K-dependent proteins in the blood (e.g., the level of carboxylated osteocalcin or MGP) as shown in Figure 3.
Passwater: How much oral supplementation is required to produce this optimal level?
Schurgers: Several population-based studies have investigated the beneficial health effects of high vitamin K intake. Some studies have only focused on vitamin K1, but in all studies in which K1 and K2 were measured separately, the health effects of K2 were more prominent than those of K1. Moreover, the limited data available suggest that the “long-chain” menaquinones MK-7, MK-8 and MK-9 are the most active compounds for health effects beyond haemostasis.
Well-documented examples are the prevention of cardiovascular disease and the prevention of cancer. In all published population surveys, the maximal health benefit was obtained in the highest tertile or quartile for vitamin K2 intake with an average vitamin K2 intake of 45 micrograms per day. That is the reason why tablets or capsules containing 45 mcg/day are most ideally marketed. This dose is 50% of the recommended intake for vitamin K, so it falls within the definitions for a food supplement.
The obvious question is whether this dose provides optimal benefit. The recent clinical trial performed on healthy postmenopausal woman showed the health benefits of 180 mcg/day.
Passwater: Over what time period should vitamin K2 be given to result in a detectable clinical effect?
Schurgers: In healthy volunteers, a biochemical effect (i.e., improved activation of the vascular calcification inhibitor MGP) is seen within six weeks, with a maximal effect after three months. Clinical benefits take more time to be achieved; measurable bone and cardiovascular benefits in a clinical trial (MenaQ7 over placebo) became visible after at least one year, with a more pronounced effect after two and three years.
Passwater: The Japanese consume a lot of natto and thus vitamin K2 in the form of MK7. How do the Japanese people do in terms of osteoporosis and longevity?
Schurgers: Soyfoods have long been a part of traditional Asian diets; they provide plentiful amounts of high-quality protein and have a favorable fatty acid profile. Moreover, fermented soybeans (i.e., natto), a traditional Japanese food, contain more than 100 times as much vitamin K2 as various cheeses and promote gamma-glutamylcarboxylation (i.e., activation of vitamin K-dependent proteins).
Increasing evidence indicates a significant role for vitamin K in bone metabolism. Scientists found a large geographic difference in serum vitamin K2 (menaquinone-7; MK-7) levels in postmenopausal women in Japan. Serum concentrations of MK-7 were significantly higher in frequent natto eaters, and natto intake resulted in a marked and sustained increase in serum MK-7 concentration. A statistically significant inverse correlation was found in women between the incidence of hip fractures and natto consumption in each prefecture throughout Japan. These findings indicate that the large geographic difference in MK-7 levels may be attributable, at least in part, to natto intake and  suggest the possibility that higher MK-7 level resulting from natto consumption contribute to the relatively lower fracture risk in Japanese women.(3).
suggest the possibility that higher MK-7 level resulting from natto consumption contribute to the relatively lower fracture risk in Japanese women.(3).
Passwater: Well, Dr. Schurgers, once again it has been fascinating chatting with you about the newest research on vitamin K2. Vitamin K2 provides us with significant health benefits in more ways than we ever imagined a few years ago. Please keep up your great research. WF
Dr. Richard Passwater is the author of more than 45 books and 500 articles on nutrition. Dr. Passwater has been WholeFoods Magazine’s science editor and author of this column since 1984. More information is available on his Web site, www.drpasswater.com.
References
1. A.C. Allison, “The Possible role of Vitamin K Deficiency in the Pathogenesis of Alzheimer’s Disease and in Augmenting Brain Damage Associated with Cardiovascular Disease,” Med Hypotheses. 57 (2), 151–155 (2001).
2. L.J. Schurgers et al., “Vitamin K-Containing Dietary Supplements: Comparison of Synthetic Vitamin K1 and Natto-Derived Menaquinone-7,” Blood 109 (8), 3279–3283 (2007).
3. M. Kaneki et al., “Japanese Fermented Soybean Food as the Major Determinant of the Large Geographic Difference in Circulating Levels of Vitamin K2: Possible Implications for Hip-Fracture Risk,” Nutr. 17 (4), 315–321 (2001).
Published in WholeFoods Magazine, August 2014










