What are tocotrienols and why should you care? Let’s look at some of the recent news about tocotrienols, a form of vitamin E that involves the prevention of cancer, heart disease and inflammation, as well as being important to nerve health and protective against radiation and bacteria. We’ll also discuss whether the form of vitamin E in most dietary supplements is the primary form of vitamin E in American diets, and if not, can it matter?
The first 90 years of vitamin E research produced prolific and notable discoveries, while recent studies are providing a wealth of new information. Until the last few decades, attention has focused mostly on the biological activities and underlying mechanisms of the vitamin E form called “alpha-tocopherol.” Alpha-tocopherol is the vitamin E primarily found in supplements, but is not the primary form of vitamin E found in the American diet. Alpha-tocopherol is only one of more than eight natural compounds that have vitamin E activity. That’s a key point—but, just what is “vitamin E activity” anyway? We’ll examine what is meant by “vitamin E activity” later. First, let’s look at some exciting news about the tocotrienol family of vitamin E compounds.
There are two main families of compounds that  have vitamin E activity: the “tocopherol” family and the “tocotrienol” family. There is also a lesser-known family called tocomonoenols. To make things even more confusing, the two main family names sound alike and are spelled similarly. A quick distinction is that one family has a “p” in its name and the other family has a “t” in the middle. I would prefer to have them known as vitamin E1 and vitamin E2. We make similar distinctions for vitamins K1 and K2 and for vitamins D2 and D3. In my humble opinion, calling the two vitamin E families vitamins E1 and E2 would be much clearer as well as simpler.
have vitamin E activity: the “tocopherol” family and the “tocotrienol” family. There is also a lesser-known family called tocomonoenols. To make things even more confusing, the two main family names sound alike and are spelled similarly. A quick distinction is that one family has a “p” in its name and the other family has a “t” in the middle. I would prefer to have them known as vitamin E1 and vitamin E2. We make similar distinctions for vitamins K1 and K2 and for vitamins D2 and D3. In my humble opinion, calling the two vitamin E families vitamins E1 and E2 would be much clearer as well as simpler.
It is important to know that even though they all share the biochemical actions that qualify them to be called vitamin E, the various forms have unique properties that result from their differences in chemical structure. The primary chemical difference between the tocopherol and tocotrienol families is that tocotrienols have double bonds in their tails. We’ll talk about other differences between the alpha-, beta-, delta- and gamma-forms later.
Compounds with similar structures that have the same biological activity are called “vitamers.” Vitamers are usually, but not always, “isomers.” Isomers are basically compounds having the same elements in the same numbers (molecular formulas), but are arranged differently (structural formulas).
The discovery of tocotrienols in 1965 came much later than tocopherols (in 1922), but existed for almost 20 years as an obscure vitamin E. I mentioned that there are eight natural forms of vitamin E; four are tocopherols and four are tocotrienols. I would be remiss if I didn’t point out that synthetic vitamin E is different than natural vitamin E. When synthetic vitamin E was first manufactured, the resulting product contained equal amounts of two isomers of alpha-tocopherol. One isomer was bio-identical to natural alpha-tocopherol whereas the other isomer was foreign to nature. At that time, the natural form of alpha-tocopherol was designated as “d-alpha-tocopherol” and the un-natural form was designated as “l-alpha-tocopherol.” The synthetic mixture was called “dl-alpha-tocopherol.”
 The problem with synthetic vitamin E is exacerbated today as modern production of synthetic vitamin E produces eight stereoisomers of alpha-tocopherol in equal amounts. This results in one bio-identical form and seven stereoisomer forms that had never before appeared in nature. Today, the form of alpha-tocopherol found in nature is designated as “RRR-alpha-tocopherol,” whereas the synthetic form is better known as “all-rac-alpha-tocopherol.” Tocotrienols, on the other hand, are found only as natural compounds and are not available commercially as synthetics.
The problem with synthetic vitamin E is exacerbated today as modern production of synthetic vitamin E produces eight stereoisomers of alpha-tocopherol in equal amounts. This results in one bio-identical form and seven stereoisomer forms that had never before appeared in nature. Today, the form of alpha-tocopherol found in nature is designated as “RRR-alpha-tocopherol,” whereas the synthetic form is better known as “all-rac-alpha-tocopherol.” Tocotrienols, on the other hand, are found only as natural compounds and are not available commercially as synthetics.
Tocotrienols have proven to contain some exceptional properties that are not shared by their “older” tocopherol siblings. Although both tocopherols and tocotrienols are powerful antioxidants—known as early as 1937 for food protection—today the brightest spot for tocotrienol research is in cancer and cardiovascular disease. In Part Two of this series, we will discuss why tocotrienols are more powerful antioxidants than the tocopherols.
Barrie Tan, Ph.D., discussed tocotrienol health benefits with us in 2008, and now is back to update us on the emergent, promising fields of tocotrienol research, including angiogenesis, bioavailability, inflammation, antioxidant properties, metabolic syndrome and obesity, radiation protection, tocopherol interference, and the recently discovered effect on cognitive impairment. He will also expand on the two known areas of tocotrienol research, namely cancer and cardiovascular disease.
Dr. Tan earned his doctorate in chemistry with an emphasis on biochemistry at the University of Otago, New Zealand. He later became a professor of chemistry and food science/nutrition at the University of Massachusetts, Amherst. His research expertise includes lipid-soluble materials such as carotenoids, tocotrienols/tocopherols, CoQ10, omega-3s and cholesterol. He was the first to introduce tocotrienol’s benefits to the nutrition industry. He founded American River Nutrition Inc. in 1998 and developed the first-ever tocopherol-free tocotrienol product derived from annatto beans. Today, the focus of his research is on lipid-soluble nutrients that have an impact on chronic and degenerative conditions. Dr. Tan was elected to be the senior editor of a compilation with broad focus on tocotrienol, Tocotrienols: Vitamin E Beyond Tocopherols (AOCS & CRC Press; Fall 2012), and was the organizer of the 2nd International Tocotrienol Symposium in conjunction with the 103rd Annual Meeting of the American Oil Chemists’ Society (Long Beach, CA; April 29, 2012).
Passwater: Dr. Tan, for our new readers, could you review your history with vitamin E tocotrienols?

|
|
| Figure 1: Annatto flowers (Bixa orellana). |
Tan: My first acquaintance with vitamin E tocotrienols was during my biochemical research with carotenoids as a professor at the University of Massachusetts, Amherst. At the time, I was studying palm carotenes, and noticed a nearly colorless oil, which we later identified to be largely tocotrienols. The discovery was followed by a patent on the commercial extraction process and my founding of Carotech in 1988, which today is a major supplier of palm-derived products, including tocotrienols. After the sale of Carotech in the early 90s, I continued to explore tocotrienol extraction from rice bran, a technology later adopted by Eastman-Kodak. As did Sean Connery in the Medicine Man movie (1992), I stumbled onto my next discovery: the unique source of tocotrienols in the South American annatto plant with its fragile, yet beautiful pastel white or pinkish flowers and characteristic bundle-like filaments containing anthers (see Figure 1).
It takes approximately a month for the annatto plant to see its robust buds become immature greenish pods. The plant was named Bixa orellana after the Spanish explorer Francisco de Orellana, who discovered it in the Amazonia in the 1500s. The British later nicknamed this the “lipstick” plant because of the intense maroon color. It was first introduced to the United States as a food colorant about 150 years ago.
It takes another two months for the tiny pods to mature and grow to larger ones, about the size of figs. They graduate from stunning green, yellow, to red pods, and finally open when fully ripe to reveal the annatto beans (see Figure 2). These beans contain the most powerful vitamin E antioxidants of Amazonia known to date. Annatto remains the first and only true source of nature-derived vitamin E that supplies tocotrienols without tocopherols.

|
|
| Figure 2: Annatto pods (“Lipstick” tree). |
Passwater: So, the main natural sources of tocotrienols are palm, rice and annatto?
Tan: Yes, but the tocotrienol composition in these sources varies. Palm and rice tocotrienol sources typically contain 25–50% tocopherols—mainly as alpha-tocopherol—while annatto is tocopherol-free vitamin E and solely composed of delta- and gamma-tocotrienol (see Figure 3).
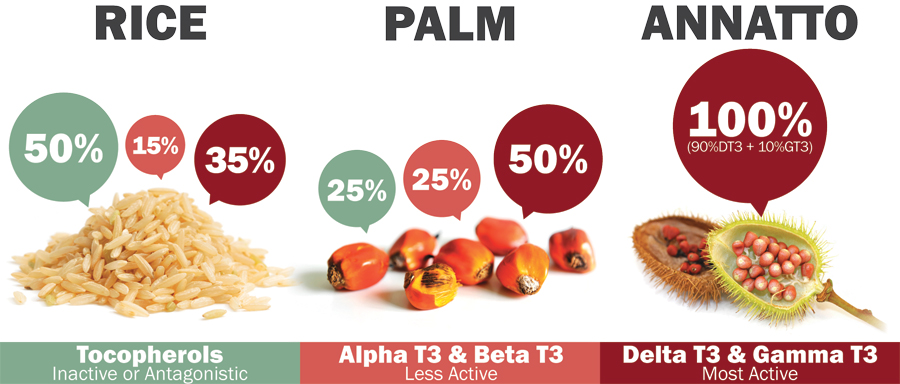
|
|
| Figure 3: Sources and composition of natural tocotrienols. |
Passwater: The richest sources of tocotrienols seem to be tropical plants and rice. What are the richest sources of tocotrienols in North American and European diets? Similarly, which food item provides the most tocotrienols to Western diets?
Tan: Aside from the major sources, tocotrienols can be found in oils and fats such as wheat bran, wheat germ, corn, oat, grapeseed, coconut, barley, margarine and lard. Vegetables and grains such as carrots, cauliflower, broccoli, peas, wheat, barley, oat, rice and rye, and fruits including avocados, apricots, blueberries, black currants, grapes and olives also contain tocotrienols. Almond, cashew, coconut, macademia and pistachio are nuts that contain tocotrienols, while they can also be found in certain meats and eggs (1).
Passwater: How much tocotrienol is in a typical American diet? How much should there be?
 Tan: Higher tocotrienol consumption from foods is desirable, but not realistic in the United States. Based on 2,000 calories, the U.S. diet contains disappointingly low amounts of tocotrienol, maybe about 2–3 mg/day. Some of this tocotrienol content may be found in less healthful foods such as macaroni, Danishes, donuts, rolls and snacks that include candy bars, cookies and potato chips (1). North Americans consume some 70% of vitamin E as gamma-tocopherol because of the abundance of soy- and corn-derived products in the diet (2). Vitamin E intake can be obtained from the USDA database (3). U.S. daily dietary consumption of vitamin Es may be broken down to about 18 mg gamma-tocopherol (4), 10–12 mg alpha-tocopherol, and 7–10 mg of all other tocols. (Tocol is the name for the two-ring structure [benzopyran-6-ol] basic to all vitamin E compounds.) Tocotrienol consumption from food is much greater in Japanese and Southeast Asian populations, where rice bran oil or palm oil is a regular part of the diet. In parts of South America, tocotrienol consumption is also very high, since annatto extracts are used in food preparation.
Tan: Higher tocotrienol consumption from foods is desirable, but not realistic in the United States. Based on 2,000 calories, the U.S. diet contains disappointingly low amounts of tocotrienol, maybe about 2–3 mg/day. Some of this tocotrienol content may be found in less healthful foods such as macaroni, Danishes, donuts, rolls and snacks that include candy bars, cookies and potato chips (1). North Americans consume some 70% of vitamin E as gamma-tocopherol because of the abundance of soy- and corn-derived products in the diet (2). Vitamin E intake can be obtained from the USDA database (3). U.S. daily dietary consumption of vitamin Es may be broken down to about 18 mg gamma-tocopherol (4), 10–12 mg alpha-tocopherol, and 7–10 mg of all other tocols. (Tocol is the name for the two-ring structure [benzopyran-6-ol] basic to all vitamin E compounds.) Tocotrienol consumption from food is much greater in Japanese and Southeast Asian populations, where rice bran oil or palm oil is a regular part of the diet. In parts of South America, tocotrienol consumption is also very high, since annatto extracts are used in food preparation.
Passwater: What is new in tocotrienol research since our last interview in 2008?
Tan: Currently, tocotrienols have reached a new measure of research height: more than one-third of all vitamin E tocotrienol research of the last 30 years was published in the last three years (2009–2011)! Of these publications, the limelight is on cancer research. Tocotrienols—but not tocopherols and, in particular, not alpha-tocopherol—have repeatedly been shown to inhibit proliferation and induce cancer cell death, and cells with the greatest degree of malignancy are most sensitive to the apoptotic action of tocotrienol (5–7).
Passwater: These are very important points, but this may be too much biochemistry to spring on some of our non-biochemist readers. We have repeatedly mentioned the alpha-tocopherol form of vitamin E. This is the form most often found in dietary supplements. Yet, the gamma-tocopherol form of vitamin E is more plentiful in the American diet than the alpha-tocopherol form. Gamma–tocopherol-rich foods include corn oil, soybean oil, margarine and dressings. Alpha–tocopherol-rich foods include wheat germ oil, sunflower and safflower oils. Please explain to our readers what we are talking about with the classifications “alpha-“, “beta-,“ “gamma-“ and “delta-“ in terms of both families of vitamin E.
Tan: “Alpha-“, “beta-“, “gamma-“ and “delta-“ are designations for the four types of tocopherols and four types of tocotrienols that have the same number of elements, but are arranged differently (i.e., they are isomers). If you imagine vitamin E as molecules with heads and tails (almost like a tadpole), the alpha-, beta-, gamma- and delta-designations are based on the bulkiness of the molecule’s head region (see Figure 4). Alpha-tocopherol and alpha–tocotrienol have the bulkiest heads with three methyl groups each, while beta- and gamma-tocopherol and -tocotrienol are less bulky with two methyl groups; delta-tocopherol and -tocotrienol are the least bulky molecules with only one methyl group. The less bulky the molecule, the greater the activity.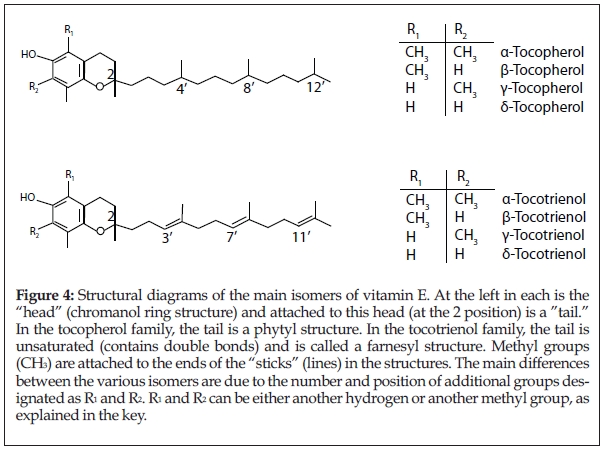
Passwater: It appears to me that in nature, delta-tocotrienol is the first vitamin E formed in plants, with methylation (adding methyl groups) leading to the other tocotrienols and hydrogenation (adding hydrogen) producing the respective tocopherols. There are more and more reports about gamma-tocopherol preventing cancer and alpha-tocopherol being ineffective.
Tan: True. The most recent one is the report by a group at Rutgers University, in which gamma- and delta-tocopherol—but not alpha-tocopherol—were found to have cancer preventive activity (8). In addition, a follow-up of a large-scale clinical trial on prostate cancer, entitled the Selenium and Vitamin E Cancer Prevention Trial (SELECT), claimed that alpha-tocopherol supplementation increased prostate cancer risk by 17% (9). Gamma-tocopherol in prostate cancer, on the other hand, had previously been shown to have anti-cancer properties (10, 11). Likewise, delta- and gamma-tocotrienols appear to have a much greater effect on various cancers—including those of the breast, prostate, pancreas, colon, lung and skin—than alpha-tocotrienol, which is largely ineffective in cancer applications.
Passwater: What is it about the chemical structure of delta- and gamma-tocopherols and tocotrienols that make them more anti-carcinogenic than the alpha-forms of vitamin E?
Tan: One reason involves the structure of vitamin E molecules, which is extremely important to their function. The alpha-forms of vitamin E have a much bulkier head, which is due to three extra methyl groups that are attached to the molecule. Gamma-forms of vitamin E have two extra methyl groups, while delta-forms have only one extra methyl group. Delta- and gamma-forms of vitamin E are also simply called “desmethyl” vitamin E due to the lack of an extra methyl group in the molecule’s head region.
Passwater: While on the subject of structural differences and function, earlier, we  mentioned that the difference between the tocopherols and tocotrienols is that the “tail” of tocotrienols is unsaturated and has a double bond, whereas the tail of tocopherols is saturated and has no double bonds (see Figure 4). What functional advantage does this impart to tocotrienols?
mentioned that the difference between the tocopherols and tocotrienols is that the “tail” of tocotrienols is unsaturated and has a double bond, whereas the tail of tocopherols is saturated and has no double bonds (see Figure 4). What functional advantage does this impart to tocotrienols?
Tan: The vitamin E tail anchors into lipid membranes constituting the cell wall. Because the membrane is made of fatty acids and oftentimes unsaturated fats—such as the beneficial omega-3s (as found in the eye and brain, for example)—cell walls of unstable lipids need a lot of oxidation protection. Within these membranes is a great space for vitamin E to be. They police radical oxidants to prevent cell membrane damage.
While both tocopherols and tocotrienols have the same structural heads, tocotrienols’ unsaturated tail is shorter and more flexible, enabling them to move freely through the cell membrane to cover larger areas for added protection. The smaller tocotrienol molecule with its shorter tail zigzags through the cell membrane faster and with greater agility, while the larger tocopherol with its longer tail staggers through the membrane more slowly and without cellular suppleness.
Passwater: It is common for a single nutrient to have more than one function and yet be most widely known for a single function. As an example, vitamin D is known for preventing rickets, but it is important for more than 200 different functions other than bone health. Today, vitamin E is best known for its antioxidant functions. However, vitamin E’s antioxidant activity may not be its most important biochemical function. Recent research points to cell signaling, enzymatic activities, gene expression and neurological functions as possibly being more important.
For decades, vitamin E was called the “anti-sterility” vitamin because of its role that led to its discovery. This was before antioxidant activity was known. In 1922, Drs. Herbert Evans and Katherine Bishop discovered that when “vitamin E” was missing from the diet of laboratory rats, they became infertile. The vitamin E active factor was first purified and extracted from wheat germ oil in 1936 and its chemical structure elucidated in 1938. Vitamin E was then given the chemical name “tocopherol” from the Greek words “tocos” (meaning childbirth) and “ferein” (to bring forth). The “ol” was added to indicate that the compound contained a hydroxyl (OH) group. It is important to assign accurate chemical names to vitamins so that we know exactly which molecule we are studying rather than using an umbrella term such as ”vitamin E.” It wasn’t until 1968 that the Food and Nutrition Boards of the National Academy of Sciences officially recognized vitamin E as an essential nutrient for humans.
When it was later discovered that there were several related compounds that had the ability to prevent animals from becoming infertile, they were ranked in comparison to alpha-tocopherol. Due to the differences in activity of the various forms of vitamin E, the dosages should be expressed in terms of International Units (IU). By definition, one IU for vitamin E is the biological equivalent of about 0.667 mg d-alpha-tocopherol (2/3 mg exactly). For some reason, when labeling vitamin E dosage in dietary supplements, vitamin E activity is reported for only the alpha-tocopherol form. The other vitamers are considered to be equal to zero. Why?
An old 1965 vitamin E activity rating system, based on biological activity in rat gestation resorption, rated beta-tocopherol as being 50% as effective as alpha-tocopherol, gamma-tocopherol at 10% as effective and delta-tocopherol at 3% as effective as alpha-tocopherol. The catch is that these efficiency rankings are according to rat gestation resorption and not according to anti-cancer activity. This antiquated rating system set the IU standards still in use today. The use of vitamin E IU is rendered meaningless for humans, since the IU was based on fetal resorption in rats. Also, only half of the population—females—would benefit from fetal resorption prevention. Although the U.S. Food and Drug Administration requires the use of IU for alpha-tocopherol only, this currency has long expired. The new currency of vitamin E should simply be mg, not the outdated IU.
In the original biological measurement of rat gestation resorption, a virgin rat is made vitamin E-deficient and then is impregnated. Next, the number of fetuses that are resorbed is determined. Then, you take other rats that have been fed various amounts of the form (vitamer) of vitamin E you are testing and do the same thing to see what is the lowest dose needed to prevent fetal resorption. As vitamin E expert Dr. Maret Traber remarked in a 1997 interview in this column, “In this day and age, I wish we could say we have some great molecular biology answer, but that’s the standard for determining bioavailability, believe it or not.” This bioassay developed by Drs. M. Joffe and P.L. Harris in 1943 is relative only to fertility, not all of the functions that we are looking for when we take vitamin E supplements. Is rat gestation resorption a relative indicator of what we are looking for when we take vitamin E as an antioxidant? Is rat gestation resorption strictly an antioxidant function? Is rat gestation resorption an indicator of anti-cancer action?
In Part 2 of this series, we will rank the antioxidant activity of the various tocopherols and tocotrienols.
Dr. Tan, before I interrupted you, you mentioned that tocotrienol forms of vitamin E produced the most apoptotic action against cancer cells. Not all of our readers are familiar with the role of apoptosis. Would you please explain what apoptosis is and why that is important for killing cancer cells?
Tan: Apoptosis is also called “programmed cell death.” It is essential in embryo development and fetus growth. If apoptosis is defective, uncontrolled cell proliferation can lead to cancer, as well as autoimmune and inflammatory diseases.
Passwater: Are there many studies that show tocotrienols are effective against cancer cells?
Tan: In cell line studies, tocotrienols seem to kill all cancer cells studied, which would be too much to cover here. Just in animal studies alone, cancer inhibition by tocotrienol has been shown for breast, prostate, pancreas, skin, lung, liver and colon. Without exception, delta- and gamma-tocotrienol were the two most potent vitamin E isomers. Interestingly, whenever tocopherols were tested, the delta-isomer was the most potent tocopherol in inhibiting breast cancer and sphingolipid synthesis in human cancer cells in vitro (12, 13) and lung tumorigenesis in vivo (14). So, there is something to be said about the “delta” structure of vitamin E.
Passwater: What are some of the mechanisms at work here?
Tan: Basically, there are several mechanisms involved including antioxidant mechanisms, gene interaction mechanisms, apoptosis and blood vessel formation.

|
| Barrie Tan, Ph.D. |
Passwater: Okay, what do these mean in lay terms? These concepts are important, but they are only useful if people can understand them. Most of our readers understand antioxidant mechanisms, but the general public has only a vague idea about what antioxidants are and what they do. The public sees the word “antioxidant” all the time, but studies show the general public, as a rule, is not able to explain what antioxidants are and what they do. Likewise, the term “gene interaction mechanisms” may be vague to many readers.
Tan: First, please allow me to answer this technically and then I will translate it into plain English. Some researchers attribute the anti-cancer effects to tocotrienols’ antioxidant activity (15), downregulating and/or degrading HMG CoA reductase, the enzyme responsible for cholesterol production in the body (16, 17), increased apoptosis (caspase-3 apoptotic pathway stimulation) (5) and vascular endothelial growth factor (VEGF) inhibition (18–20).
Okay, now for the English translation. As antioxidants, tocotrienols have been indicated to have anti-cancer properties because they can protect cells from free radical damage. Free radicals are unstable molecules, and antioxidants basically sacrifice themselves in order to stabilize the free radicals. Some scientists believe that tocotrienols affect cancer due to their ability to “dial down” the enzyme responsible for cholesterol production in the body, and cholesterol production is abnormally high in cancer cells. As mentioned earlier, tocotrienols also induce apoptosis or programmed cell death in cancer cells. They do so by activating caspase-3, a protein that is instrumental in the apoptotic pathway. Finally, tocotrienols reduce vascular endothelial growth factor, a protein involved in signaling angiogenesis, which in cancer is the aberrant growth of blood vessels.
Passwater: The concept of tocotrienol’s anti-angiogenic mechanism is fascinating. Can you explain this concept further?
Tan: Recent studies showed that tocotrienols, but not tocopherols, inhibit angiogenesis (in cancer, the aberrant and uncontrolled growth of blood vessels), an indispensable step in tumor growth beyond a 1-mm mass. To fuel the growth of a tumor, nutrients are needed. New (but aberrant) blood vessels are made to facilitate the delivery of these nutrients to the tumor. The tumor stimulates the growth of these blood vessels by a process known as “angiogenesis,” meaning the growth of new and aberrant blood vessels. Tocotrienols, particularly delta-tocotrienol, cut the nutrient lifeline to the tumor, essentially starving the tumor to death. The body can produce what is called vascular endothelial growth factor (VEGF), which regulates angiogenesis. VEGF stimulates angiogenesis when it is taken up by cells via a receptor in cell membranes. Tocotrienol downregulates the production of these VEGF receptors, therefore blocking intracellular signaling of VEGF and inhibiting angiogenesis (19).
In addition, tocotrienol inhibits the proliferation and formation of tubes (the in vitro equivalent of blood vessels) by aortic endothelial cells, where delta-tocotrienol had the strongest inhibitory activity (21). Gamma-tocotrienol was also shown to inhibit the angiogenesis of umbilical vein endothelial cells (22). Since angiogenesis is essential to tumor growth, its inhibition stunts tumor growth and prevents cancer metastasis. Interestingly, delta- and gamma-tocopherol were also the two most potent anti-angiogenic isomers of the tocopherol family (14).
Tocotrienol may well work on dual antitumor mechanisms that include the removal of the vital nutrient-to-tumor lifeline (via inhibiting angiogenesis) and the targeting of tumor cells via signals (5, 23). The fact remains that there are many compelling modes of action shown in cell lines (based on the “need to learn and know” concept), but validation is in animal studies (based on a “system phenomenon”). Perhaps we can say that a “mode of action” guides, a “system phenomenon” validates and a “human effect” proves. Tocotrienol research is following a compelling pattern. At the same time, please remember this—tocotrienol is only a humble vitamin E, which makes such activities truly remarkable.
Passwater: You said some researchers attribute the anti-cancer action of tocotrienols to those factors. Are there other actions as well?
Tan: Many more researchers compellingly proposed gene products (some up- and some down-regulated, and some very selective), cell signals, cytokines and activating factors that are responsible for the eventual path leading to cancer death brought about by tocotrienols.
Passwater: These cell line studies are beyond the scope of this interview to provide adequate coverage and do them justice. Perhaps, we can just say that researchers have invariably shown that delta- and gamma-tocotrienol were the two most potent isomers. For more details, readers are directed to the second edition of Tocotrienols: Vitamin E Beyond Tocopherols.
Are there currently any clinical studies for tocotrienol cancer research?
Tan: A phase I dose-escalation trial on delta-tocotrienol in patients with resectable pancreatic cancer is underway and so far shows no adverse effects up to 3,200 mg/day, while apoptosis of cancer cells in patients was observed at the lowest dosage of 200 mg/day (24). Further analysis of tissue specimens and a phase II trial are planned. Other cell line and animal studies undertaken by at least two independent research groups clearly underscore and lend unambiguous support to delta-tocotrienol’s effect on pancreatic cancer (25–28). Pancreatic cancer remains the most lethal of all cancers with only a 6% survival rate (about one in 20) (29), and hence this research is of enormous importance. Steve Jobs, Patrick Swayze, Luciano Pavarotti, Dizzy Gillespie and Count Basie all have died of this disease. Supreme Court justice Ruth Bader Ginsburg is living with it.
Passwater: Dr. Tan, let’s take a break and continue next month with other important health roles of tocotrienols including heart disease, chronic inflammation, nerve health, radiation protection and others. WF
Dr. Richard Passwater is the author of more than 45 books and 500 articles on nutrition. Dr. Passwater has been WholeFoods Magazine’s science editor and author of this column since 1984. More information is available on his Web site, www.drpasswater.com.
References
The complete references will follow the last part of this series.
Published in WholeFoods Magazine, July 2012










